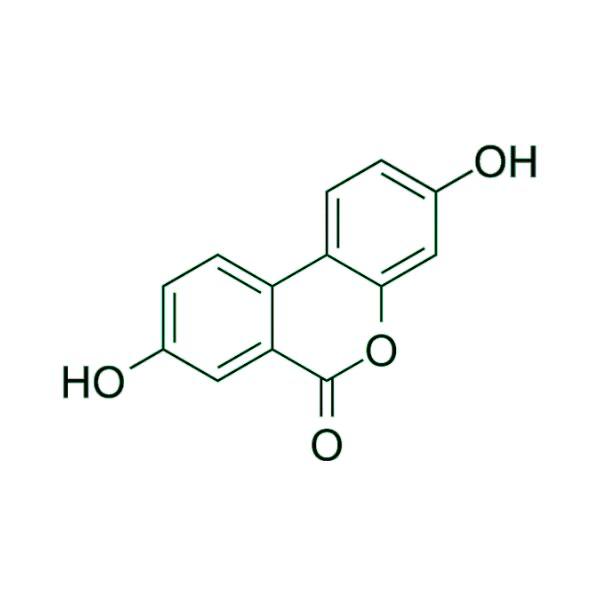
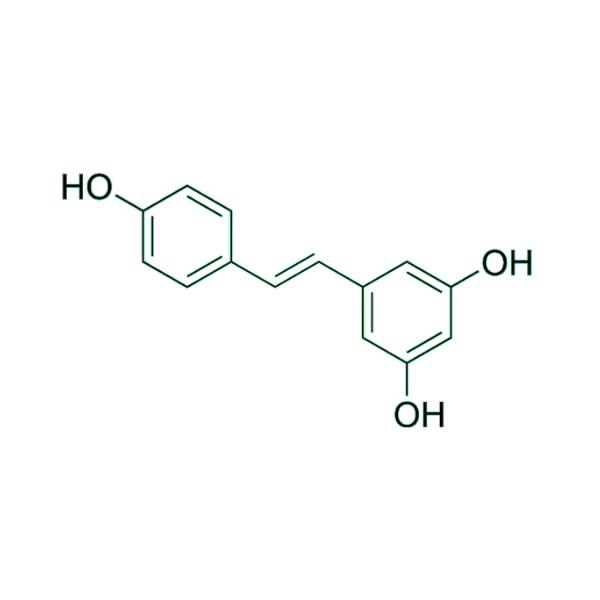
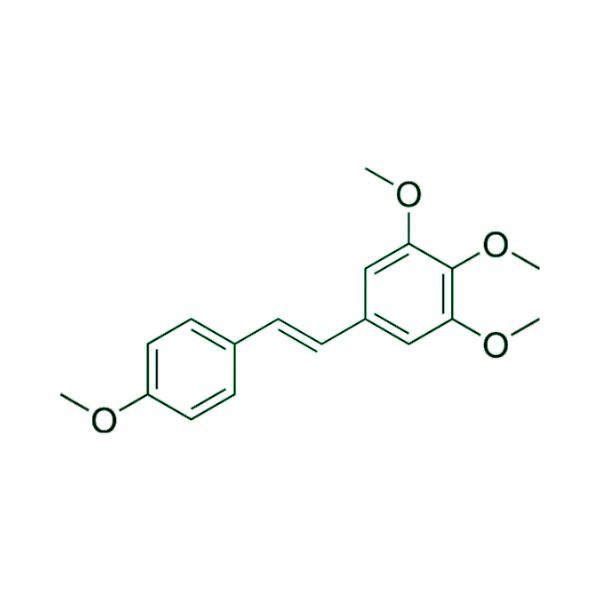
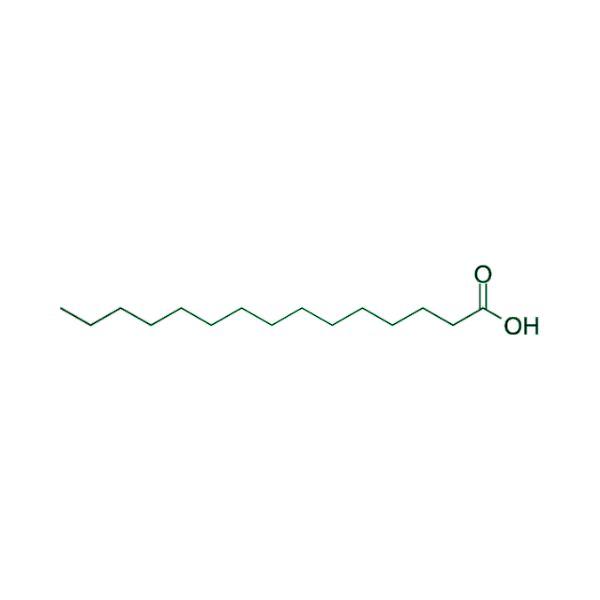
Advancing Small-Molecule Science from Discovery to Manufacture
Science-Driven API Development, Built for Scale and Compliance
Aurapha is a research-driven pharmaceutical company specializing in the development, scale-up, and manufacturing of active pharmaceutical ingredients (APIs). Built on strong scientific foundations, we translate validated molecules into reliable, high-quality drug substances for global pharmaceutical markets.
Advancing Scientifically Validated Molecules Toward Reliable Supply
Specialized Small-Molecule & Process Expertise
Small-Molecule Therapeutics
Postbiotics & Polyphenol Derivatives
Neurology & Metabolic Health Compounds
Process Chemistry & Scale-Up
Scientific Excellence Driving Reliable API Development
Chemistry-Driven API Development
We bring deep expertise in synthetic organic chemistry and process development to design robust, scalable routes for complex small-molecule APIs.
Integrated Development & Manufacturing
Our integrated approach ensures seamless transition from laboratory development to pilot and commercial-scale manufacturing, minimizing risk and timelines.
Process Robustness & Reproducibility
We prioritize reproducible, well-controlled processes that deliver consistent quality across batches and scales.
Impurity & Quality Control Expertise
Early-stage impurity identification and control are embedded into our workflows to meet global regulatory and pharmacopeial standards.
Regulatory-Ready Mindset
Processes and documentation are developed with GMP, ICH, and global regulatory expectations in mind from the outset.
Experienced Multidisciplinary Team
Our team of highly qualified chemists and biologists brings strong scientific insight and practical manufacturing experience.
Integrated. Scalable. Regulatory-Ready.
Our integrated development and manufacturing model ensures consistency, quality, and efficiency from early route design through commercial-scale production. We work closely with our partners to translate laboratory science into robust, manufacturable API processes.